Menu
Others
Health Focus, Issue 528 — Multiple Influenza Vaccine Options for Year-Round Protection
Influenza is an infectious respiratory illness caused by the influenza virus.
It exhibits clear seasonal and cyclical patterns. Although flu cases can occur year-round in Taiwan, outbreaks usually peak during the autumn and winter seasons.
For high-risk groups — including adults aged 65 and older, young children, pregnant women, individuals with weakened immune systems, and those with chronic diseases — influenza infection can lead to severe complications and may even be fatal.
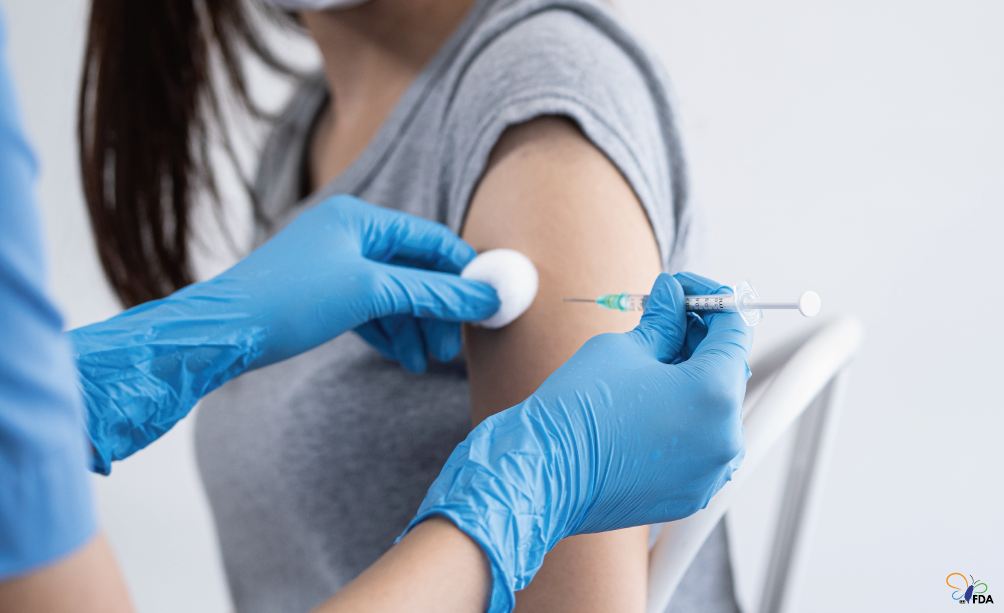
The most effective way to prevent influenza is through vaccination.
According to data from the Taiwan Centers for Disease Control (CDC), over 90% of confirmed cases and deaths occurred in individuals who had not received the current seasonal flu vaccine, underscoring the vaccine’s crucial role in reducing the risk of severe illness and death.
Diverse Vaccine Technologies, Broader Choices
Influenza vaccines can be categorized into inactivated vaccines and live attenuated vaccines. Among them, inactivated vaccines can be further classified by their production methods into egg-based vaccines and cell-based vaccines.
The egg-based process involves injecting influenza viruses into fertilized chicken eggs, where the virus replicates in the allantoic fluid. The virus is then inactivated and purified to produce the vaccine. This method offers stable production capacity but is not suitable for individuals with egg allergies.
The cell-based process, on the other hand, uses mammalian cells as host cells for virus replication. After the virus is grown, it is inactivated and purified to produce the vaccine. Because this process does not use chicken eggs, the resulting vaccine contains no egg protein residues, making it a suitable option for people with egg allergies.
A New Live Attenuated Vaccine Offers an Additional Option
This year, the Taiwan Food and Drug Administration (TFDA) approved a new live attenuated influenza vaccine (LAIV), which is administered via nasal spray. Healthcare professionals deliver the vaccine into the nasal cavity, where it is absorbed through the mucous membranes to trigger both local and systemic immune responses.
However, this vaccine is not recommended for individuals with weakened immune systems, and because it is also produced using egg-based cultivation, it is unsuitable for people with egg allergies.
With ongoing advancements and diversification in vaccine technologies, the public now has more options for flu prevention during the autumn and winter seasons.
Regardless of the vaccine type or form, the TFDA strictly monitors vaccine quality. Every batch must undergo comprehensive document review and quality testing. Only after passing inspection and receiving a Drug Release Certificate may the vaccine be distributed to medical institutions.
The public can therefore receive vaccinations with confidence, effectively enhancing protection against influenza.
Source: Taiwan Food and Drug Administration (TFDA)
Phone: 03-4227151#57270、03-2804814
Fax: 03-4272405
Email: ncu7270@ncu.edu.tw
Address: No. 300, Zhongda Rd., Zhongli District, Taoyuan City 320317, Taiwan (R.O.C.)
Views: 0
Copyright © Health Care Division, National Central University
National Central University - Health Center